
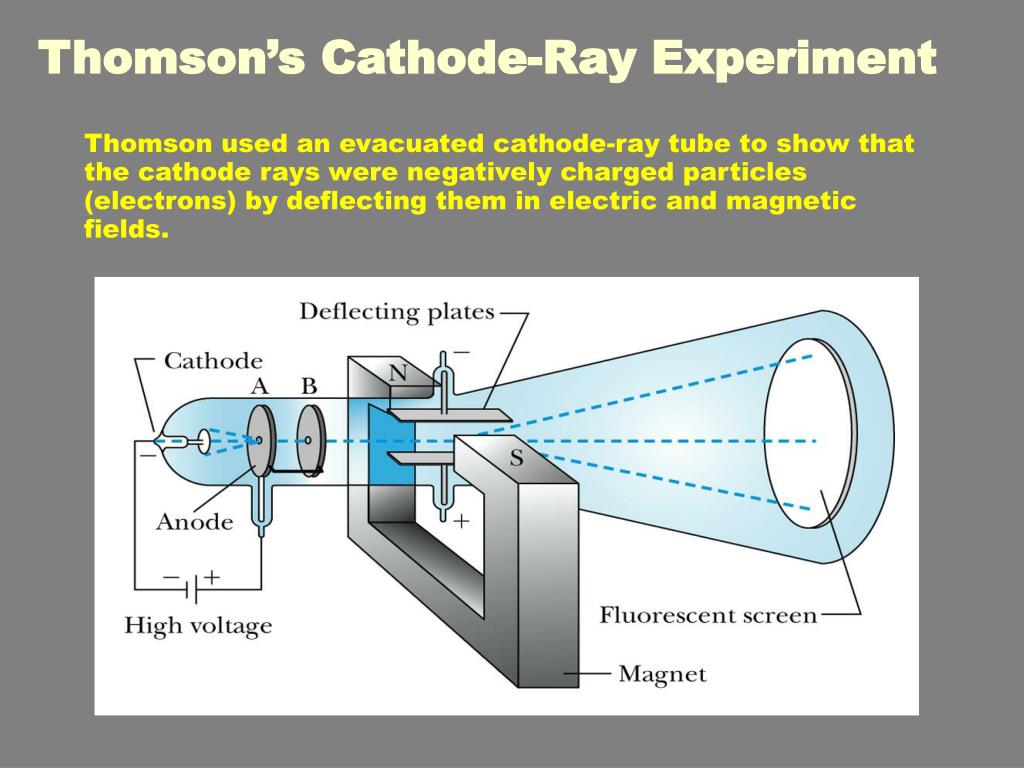
If I am able to create such a line, I may be able to generate a very basic oscilloscope that illustrates the voltages, using charged plates and probes. My end goal is to generate a straight line using a high voltage AC electrostatic field. I’m not going to be able to get any high resolution images. “I’m on the verge of getting a focused beam, and that’s when I’m going to play around with deflection, which I’m super excited about. If I do get cathode rays, it essentially lights up.” And he has managed to generate cathode rays to light up the bottom of the flask. But if you create a real vacuum, you’re not going to see anything, so I had to put a zinc sulfide layer at the bottom of the flask. “You can’t see cathode rays unless there’s air in the tube,” Rupert explained, “which generates a purplish glow because you’re creating plasma ( glow discharge formed by the passage of the electric current in a low-pressure tube). Visible cathode rays inside Rupert’s flask You’re moving the electrons fast enough to create images.” What that does is to accelerate the electrons in a direction, then you can deflect them to create pictures. “You can use either magnets ( how televisions worked, creating a magnetic field using a coil) or electrostatic fields, by placing high-voltage plates next to each other. “I’m interested in how cathode rays deflect when you put them in a magnetic field or an electrostatic field,” Rupert said.
#Cathode ray experiment definition free#
He finds whatever time he can during free blocks and study halls, and occasionally, after a hurried lunch, to immerse himself in his cathode-ray tube experiments. Hanson’s been there every step of the way, mostly to remind me about safety!” Rupert has a more rudimentary set-up in his basement at home where he can conduct some weekend research, but he’s doing the more developed experimentation in the FCS chemistry lab, under the watchful eyes of Phyllis Hanson. It’s really great to have the equipment I need available at school.” Rupert at work in the FCS chemistry lab I’m doing this in an FCS chemistry lab, using the vacuum pump that’s there. “To build my own cathode-ray tube,” he continued, “I started with a plain Erlenmeyer flask, but I eventually switched to a filtering flask, because it has a little nozzle on the side that I can pull the vacuum from. They can flow freely because there’s no air to disrupt their path.” “This creates streams of electrons, which are called cathode rays. With a cathode-ray tube, “you create a vacuum inside a glass tube, and you have super-high voltage, causing electrons to jump from the anode to the cathode,” Rupert explained. “There wasn’t one, so I decided to build my own,” he said. He emailed his chemistry teacher, Phyllis Hanson, PhD, to see if Friends’ Central had something approximating a cathode-ray tube so he could see it in action. I spent an hour down a Google rabbit hole, and I decided I wanted to try and mess with cathode-ray tubes.” “And I thought, that seems pretty straightforward, and then I got really curious about how old televisions worked. A natural engineer, he had a general understanding of the mechanism of transistors, LEDs, and LCD displays.
#Cathode ray experiment definition tv#
While watching TV one day, he found himself thinking about how it worked.

“I love understanding things,” said Rupert.
It’s a project he himself devised, and he’s exploring it with the single-minded passion and focus of the truly intellectually curious. F riends’ Central Sophomore Rupert Bowen is conducting an independent experiment building cathode-ray tubes in an FCS chemistry lab.
